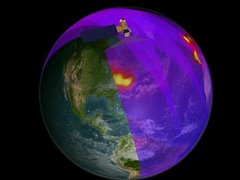
As always, Aura is monitoring stratospheric ozone levels. Yesterday, the "hole" or thinning over Antarctica reached a new low concentration - 106 Dobson units - for 2008.
Each year, ozone levels in the stratosphere drop in the Arctic and Antarctic in their respective winters. Cold temperatures in the atmosphere result in isolation of the air masses above the Arctic and Antarctic circles. This isolation, along with cold temperatures promote chemical reactions that favor the release of a chlorine atom from chemical compounds such as chlorofluorocarbons (CFCs). This Cl atom attaches itself to an oxygen atom and removes it from the ozone molecule. The result is a reduction of ozone in the ozone layer. This process is seasonal - once spring comes around and atmospheric temperatures rise, the isolated air masses return to normal circulation and the chemical reactions that caused the release of chlorine are no longer viable. Ozone levels in the stratosphere then return to normal.
Why are we so interested in stratospheric ozone hole? The ozone in the stratosphere protects all living beings on earth from harmful ultraviolet-c radiation. This uv-c causes the DNA in cells to break down, which can result in cancer. Without the ozone layer, uv-c would be able to penetrate the atmosphere and it would wreak havoc on all living things.
The Aura spacecraft also measures the other chemical players in the reactions that lead to the break down of ozone in the stratosphere in order to better understand how these reactions happen. This understanding can lead to the prevention of the release of chemicals that cause ozone loss into the atmosphere.